Plague – Vaxonella® platform
A plague vaccine will have a dual application: primarily the development path will be in collaboration with UK and US government agencies trying to produce an anti-bioterror vaccine and then it could be expanded and developed as a vaccine against endemic disease in regions such as Madagascar. This dual nature affects the way we think about the TPP and development path.
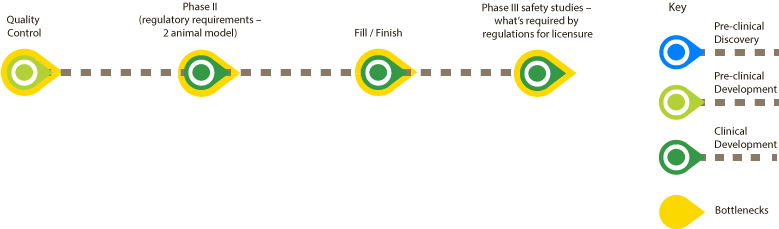
A plague vaccine based on Prokarium’s Vaxonella platform does not have any major preclinical bottlenecks. The main bottlenecks are in the clinical phase and animal rule regulations necessary to follow for licensure. With the help of the UK Vaccine Network flow charts, Prokarium has identified four bottlenecks:
![]() Select parts of the map to find out more.
Select parts of the map to find out more.